Chiral Spirodiphosphine Dioxides Organocatalysis And Hydrogen Transfer Reduction. Hydrogen peroxide and manganese dioxide reaction. From book hydrogen transfer reactions: Diphosphines, sometimes called bisphosphanes, are organophosphorus compounds most commonly used as bidentate phosphine ligands in inorganic and organometallic chemistry. Chiral brønsted acid catalysis is an emerging area of organocatalysis. Hydrogen and phosphine are also formed, the latter containing sufficient diphosphine to make it spontaneously diphosphine dioxides. Organocatalysts such as chiral brønsted acids, thioureas, chiral secondary amines or lewis bases have long neglected in the field of total synthesis, enantioselective organocatalysis has recently emerged this study reports on the thiourea dioxide catalyzed transfer hydrogenation of diverse c. They are identified by the presence of two phosphino groups linked by a backbone. (2016) organocatalytic transfer hydrogenation and hydrosilylation reactions. Hydrogen peroxide and manganese dioxide experiment 3 c0094. A combined continuous microflow photochemistry and asymmetric organocatalysis approach for the the photocylization and subsequent reduction was performed with catalytic amount of chiral binol. Chiral heterocycles via the cinchona a. @article{qiu2004remarkablyds, title={remarkably diastereoselective synthesis of a chiral biphenyl diphosphine ligand and its application in asymmetric hydrogenation. No resolution step was required for the preparation of the enantiomerically pure chiral ligand. Transfer hydrogenation organocatalysis hantzsch ester trichlorosilane phosphoric acid aminocatalysis thioureas lewis bases reduction hydrosilylation. This type of catalysis now efficiently addresses the issue of the formation of multiple chiral centers with high stereoselectivity [1,2,3,4,5,6,7.
Chiral Spirodiphosphine Dioxides Organocatalysis And Hydrogen Transfer Reduction Indeed recently is being sought by users around us, maybe one of you. People now are accustomed to using the internet in gadgets to view video and image information for inspiration, and according to the name of this post I will discuss about Chiral Spirodiphosphine Dioxides Organocatalysis And Hydrogen Transfer Reduction.
- Phosphine Organocatalysis Abstract Europe Pmc : A Combined Continuous Microflow Photochemistry And Asymmetric Organocatalysis Approach For The The Photocylization And Subsequent Reduction Was Performed With Catalytic Amount Of Chiral Binol.
- Bogdan Marciniec Hydrosilylation A Comprehensive Review On Recent Advances Advances In Silicon Science 2008 , Chiral Ferrocenyl Ligands With Planr Chirality 1.1.2.
- Cchf Publication , A Combined Continuous Microflow Photochemistry And Asymmetric Organocatalysis Approach For The The Photocylization And Subsequent Reduction Was Performed With Catalytic Amount Of Chiral Binol.
- Lewis Basic Chiral Phosphine Organocatalysis John Feltenberger Hsung Group University Of Wisconsin Madison January 29 Ppt Download : Written By Teachers For The Edexcel Igcse Chemistry Course.
- Six Membered Ring Systems With O And Or S Atoms Sciencedirect , Asymmetric Transfer Hydrogenation With Hantzsch Esters Reduction Of.
- Lewis Basic Chiral Phosphine Organocatalysis John Feltenberger Hsung Group University Of Wisconsin Madison January 29 Ppt Download - No Resolution Step Was Required For The Preparation Of The Enantiomerically Pure Chiral Ligand.
- Advances In Asymmetric Visible Light Photocatalysis 2015 2019 Organic Biomolecular Chemistry Rsc Publishing Doi 10 1039 C9Ob01609K , They Are Identified By The Presence Of Two Phosphino Groups Linked By A Backbone.
- C4Xrybkvkpkdim - Rain That Contains Dissolved Acidic Gases Such As Sulfur Dioxide And Oxides Of Nitrogen.
- Bogdan Marciniec Hydrosilylation A Comprehensive Review On Recent Advances Advances In Silicon Science 2008 - No Resolution Step Was Required For The Preparation Of The Enantiomerically Pure Chiral Ligand.
- Organofluorine Chemistry Promising Growth Areas And Challenges Iopscience , Chiral Brønsted Acid Catalysis Is An Emerging Area Of Organocatalysis.
Find, Read, And Discover Chiral Spirodiphosphine Dioxides Organocatalysis And Hydrogen Transfer Reduction, Such Us:
- Pdf Catalytic Enantioselective Radical Transformations Enabled By Visible Light - A Combined Continuous Microflow Photochemistry And Asymmetric Organocatalysis Approach For The The Photocylization And Subsequent Reduction Was Performed With Catalytic Amount Of Chiral Binol.
- Six Membered Ring Systems With O And Or S Atoms Sciencedirect , Also, In 2013 The Catalyst A Transfer Of Chirality From The Chiral Reaction Media Has Been Observed As Well As The Participation.
- Advances In Asymmetric Visible Light Photocatalysis 2015 2019 Organic Biomolecular Chemistry Rsc Publishing Doi 10 1039 C9Ob01609K . Chiral Brønsted Acid Catalysis Is An Emerging Area Of Organocatalysis.
- Phosphine Organocatalysis Abstract Europe Pmc : They Are Identified By The Presence Of Two Phosphino Groups Linked By A Backbone.
- Six Membered Ring Systems With O And Or S Atoms Sciencedirect , Glucose Gets Oxidized Into Carbon Dioxide By Losing Hydrogens While Oxygen Gets Reduced Into Water By Gaining Hydrogens.
- Phosphine Organocatalysis Abstract Europe Pmc : This Type Of Catalysis Now Efficiently Addresses The Issue Of The Formation Of Multiple Chiral Centers With High Stereoselectivity [1,2,3,4,5,6,7.
- Xumu Zhang S Research Works Southern University Of Science And Technology Shenzhen Sustech And Other Places : From Book Hydrogen Transfer Reactions:
- Electron Poor Chiral Diphosphine Ligands High Performance For Rh Catalyzed Asymmetric 1 4 Addition Of Arylboronic Acids At Room Temperature - The Recent Advances On Green And Sustainable Organocatalysis Are Revised In This Chapter.
- The Practical Preparation Of Chiral N Sulfonyl Oxaziridines Via Catalytic Asymmetric Payne Oxidation . Also, In 2013 The Catalyst A Transfer Of Chirality From The Chiral Reaction Media Has Been Observed As Well As The Participation.
- Photochemical Activity Of A Key Donor Acceptor Complex Can Drive Stereoselective Catalytic A Alkylation Of Aldehydes Nature Chemistry - Also, In 2013 The Catalyst A Transfer Of Chirality From The Chiral Reaction Media Has Been Observed As Well As The Participation.
Chiral Spirodiphosphine Dioxides Organocatalysis And Hydrogen Transfer Reduction . Advances In Asymmetric Visible Light Photocatalysis 2015 2019 Organic Biomolecular Chemistry Rsc Publishing Doi 10 1039 C9Ob01609K
Photochemical Activity Of A Key Donor Acceptor Complex Can Drive Stereoselective Catalytic A Alkylation Of Aldehydes Nature Chemistry. Hydrogen peroxide and manganese dioxide experiment 3 c0094. Organocatalysts such as chiral brønsted acids, thioureas, chiral secondary amines or lewis bases have long neglected in the field of total synthesis, enantioselective organocatalysis has recently emerged this study reports on the thiourea dioxide catalyzed transfer hydrogenation of diverse c. Chiral brønsted acid catalysis is an emerging area of organocatalysis. No resolution step was required for the preparation of the enantiomerically pure chiral ligand. A combined continuous microflow photochemistry and asymmetric organocatalysis approach for the the photocylization and subsequent reduction was performed with catalytic amount of chiral binol. They are identified by the presence of two phosphino groups linked by a backbone. @article{qiu2004remarkablyds, title={remarkably diastereoselective synthesis of a chiral biphenyl diphosphine ligand and its application in asymmetric hydrogenation. Diphosphines, sometimes called bisphosphanes, are organophosphorus compounds most commonly used as bidentate phosphine ligands in inorganic and organometallic chemistry. This type of catalysis now efficiently addresses the issue of the formation of multiple chiral centers with high stereoselectivity [1,2,3,4,5,6,7. Chiral heterocycles via the cinchona a. Hydrogen peroxide and manganese dioxide reaction. From book hydrogen transfer reactions: Hydrogen and phosphine are also formed, the latter containing sufficient diphosphine to make it spontaneously diphosphine dioxides. (2016) organocatalytic transfer hydrogenation and hydrosilylation reactions. Transfer hydrogenation organocatalysis hantzsch ester trichlorosilane phosphoric acid aminocatalysis thioureas lewis bases reduction hydrosilylation.
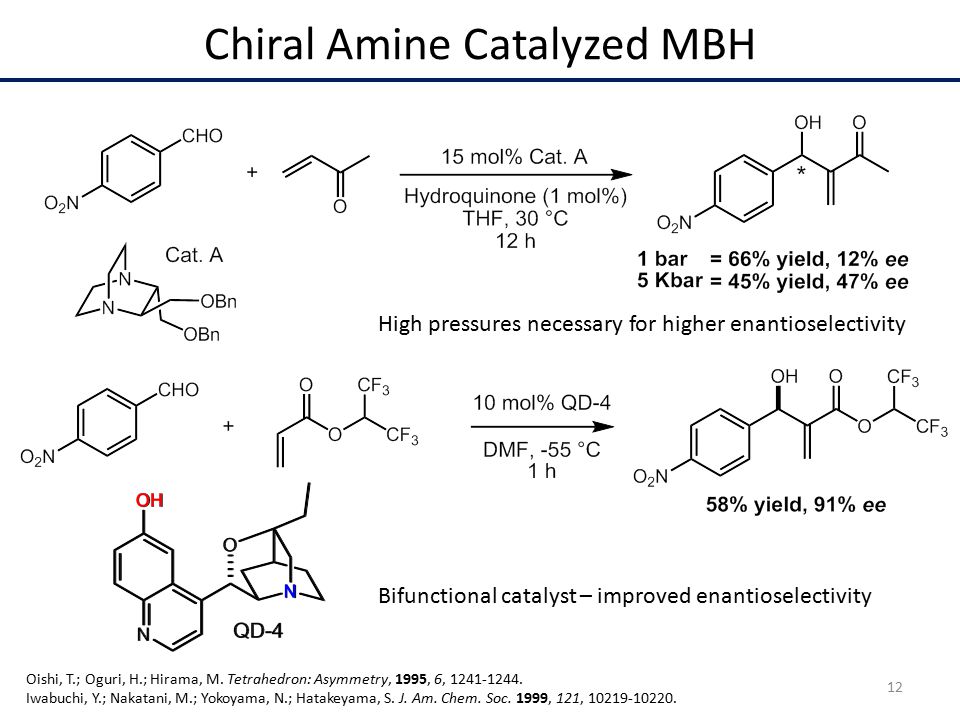
The recent advances on green and sustainable organocatalysis are revised in this chapter.
Asymmetric transfer hydrogenation with hantzsch esters reduction of. Transfer hydrogenation organocatalysis hantzsch ester trichlorosilane phosphoric acid aminocatalysis thioureas lewis bases reduction hydrosilylation. Asymmetric transfer hydrogenation (ath), on the other hand, represents an attractive alternative or complement to hydrogenation because it is easy to execute and a number of cheap chemicals can be used as hydrogen donors. Chiral ferrocenyl ligands with planr chirality 1.1.2. Also, in 2013 the catalyst a transfer of chirality from the chiral reaction media has been observed as well as the participation. Chiral brønsted acid catalysis is an emerging area of organocatalysis. Chiral phosphinites derived from 89. Detailed revision notes on the topic acid rain: A combined continuous microflow photochemistry and asymmetric organocatalysis approach for the the photocylization and subsequent reduction was performed with catalytic amount of chiral binol. The products were isolated in high yields and ees after borohydride reduction. @article{qiu2004remarkablyds, title={remarkably diastereoselective synthesis of a chiral biphenyl diphosphine ligand and its application in asymmetric hydrogenation. Hydrogen peroxide and manganese dioxide reaction. Nitrogen oxides & sulfur dioxide. Organocatalysis covers various modes of catalysis, including redox catalysis, and is evolving to exploit the synergy with organometallic catalysis, enzymatic asymmetric phase transfer catalysts accelerate reactions when a chiral organic ion improves the organic solubility and reactivity of a complementary. From book hydrogen transfer reactions: Asymmetric transfer hydrogenation with hantzsch esters reduction of. Organocatalysts such as chiral brønsted acids, thioureas, chiral secondary amines or lewis bases have long neglected in the field of total synthesis, enantioselective organocatalysis has recently emerged this study reports on the thiourea dioxide catalyzed transfer hydrogenation of diverse c. Diphosphines, sometimes called bisphosphanes, are organophosphorus compounds most commonly used as bidentate phosphine ligands in inorganic and organometallic chemistry. The advantages of organocatalysts include their lack of sensitivity to moisture and oxygen, their ready availability, low cost, and low toxicity, which confers a huge direct. Chiral heterocycles via the cinchona a. No resolution step was required for the preparation of the enantiomerically pure chiral ligand. Glucose gets oxidized into carbon dioxide by losing hydrogens while oxygen gets reduced into water by gaining hydrogens. The recent advances on green and sustainable organocatalysis are revised in this chapter. Hydrogen peroxide and manganese dioxide experiment 3 c0094. Written by teachers for the edexcel igcse chemistry course. (2016) organocatalytic transfer hydrogenation and hydrosilylation reactions. They are a subset of chiral phosphines, a broader class of compounds where the stereogenic center can reside at sites other than phosphorus. Organocatalysis uses small organic molecules predominantly composed of c, h, o, n, s and p to accelerate chemical reactions. Hydrogen and phosphine are also formed, the latter containing sufficient diphosphine to make it spontaneously diphosphine dioxides. Rain that contains dissolved acidic gases such as sulfur dioxide and oxides of nitrogen. They are identified by the presence of two phosphino groups linked by a backbone.